So I've been pondering some things back and forth. Talking with ouro in chat and seeing a few threads here and there.
I found a journal article
A Low-Cost Differential Fluorimeter for the Detection and Determination of LSD in Illicit Preparations
Philippe Baudot
I really am not impressed with the design used here but I guess that's beside the point. The article states
So I got to thinking and I came up with a rough idea for how to do some detection here. In the case of LSD, which is a pressing issue for many people taking such these days. 25I-NBOME's, DOI/DOB/DOC, bunk tabs, you name it its all out there and being passed around.
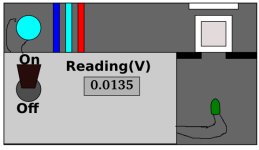
Here's the rough sketch in my head... For a dirt-cheap fluorimeter

Figure #1
Inside of a dark box:
A light source maybe a UV-A pen-lamp, maybe a small mercury vapor lamp. This needs to provide the emission source(323nm and/or 325nm). This shines through a cuvette. A cheap cuvette could actually be used to our advantage to block UV light below these desired wavelengths according to "figure #1" .
.
Now perpendicular to this light source is where the emission will be collected. Here's the trick up my sleeve. A reversed LED and an op amp type circuit. LEDs with peak wavelengths of say 500nm are cheap and widely available. Wire this up to a decent micro-ammeter, and well I haven't exactly figured the rest out yet. IE: calibration, will this actually work?, you know the fine details...
I found a journal article
A Low-Cost Differential Fluorimeter for the Detection and Determination of LSD in Illicit Preparations
Philippe Baudot
I really am not impressed with the design used here but I guess that's beside the point. The article states
In the case of LSD, fluorescence occurs between 400 and 530 nm, depending on the solvent and the pH employed. For example, excitation at 323 nm (kex max) in aqueous medium gives a blue fluorescence at 432 nm (kem max), whereas excitation at 325 nm fhex max) in aqueous molar sodium hydroxide produces a green fluorescence at 502 nm (hem max) (6,7). Observation with the eye can thus be carried out directly.
So I got to thinking and I came up with a rough idea for how to do some detection here. In the case of LSD, which is a pressing issue for many people taking such these days. 25I-NBOME's, DOI/DOB/DOC, bunk tabs, you name it its all out there and being passed around.
Here's the rough sketch in my head... For a dirt-cheap fluorimeter

Figure #1
Inside of a dark box:
A light source maybe a UV-A pen-lamp, maybe a small mercury vapor lamp. This needs to provide the emission source(323nm and/or 325nm). This shines through a cuvette. A cheap cuvette could actually be used to our advantage to block UV light below these desired wavelengths according to "figure #1"
Now perpendicular to this light source is where the emission will be collected. Here's the trick up my sleeve. A reversed LED and an op amp type circuit. LEDs with peak wavelengths of say 500nm are cheap and widely available. Wire this up to a decent micro-ammeter, and well I haven't exactly figured the rest out yet. IE: calibration, will this actually work?, you know the fine details...