So one happy weekend a couple of weeks ago, Infundibulum, The Traveler and I were hanging out here together and we did a little experiment:
Pre-work:
Harmalas were extracted from syrian rue, precipitated mostly pure harmine (1 ) by adding sodium bicarbonate until no more color change was seen. This was left to stand for an hour, filtered to retrieve harmine, and then some sodium carbonate was added till no color change was noticed anymore, to precipitate a mixture of harmine/harmaline (2). This mixture (2) was filtered/dried and redissolved in acidic solution. To this solution, sodium bicarb was added to re-precipitate the harmine from mixture, which was filtered, and then sodium carb was added to precipitate reasonably pure harmaline (95%).
The reduction:
We dissolved the harmaline in some cheap supermarket vinegar, added excess zinc dust (which had been kept in a plastic bag for 2 years, we were affraid it had mostly oxidized and would not work). A lot of zinc dust just stayed on the bottom, and we could notice some very very fine bubbles in the vinegar (which is Hydrogen gas that is formed). We stirred occasionally and let it sit for a couple of hours. Finally, solution was filtered and sodium carbonate added. There was a lot of precipitation.
The precipitation was a mixture of alkaloids as well as zinc carbonate. In literature, this reduction is often done with HCl instead of vinegar, and then adding ammonium chloride, and later ammonium hydroxide to precipitate the base alkaloids. Apparently ammonium chloride prevents zinc salts from precipitating, so you only have your THH. But since we didnt really consider consuming and it was just a test, we precipitated with sodium carbonate and had excess zinc carbonate.
Zinc carbonate is NOT soluble in alcohols, so theoretically someone could use some ethanol to soak the alkaloid+zinccarbonate mixture and evap that to have alkaloids free of the zinc carb.. To analyze, after filtering this mixture, I soaked it in methanol, which was inserted in
The results:

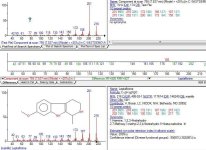
GC-MS results show the conversion from harmaline to THH to be virtually 100%. There are no traces of harmaline detected, only small peak of harmine left (zinc is probably not strong enough to reduce harmine to harmaline but it might be it does, I cannot quantify that peak with the program I have it doesnt "detect" it).
There are other small peaks which are very likely the vinegar impurities (acetic compounds... one that seems to be glycerol acetate and other long chain compounds which seem to be carboxylic acids), Im not sure if any of these compounds were created from reduction of vinegar or if they are present in vinegar in the first place, but they are certainly not coming from the harmalas. There is one other small peak which is a phthalate, coming from plastic container used during dissolution of the alkaloids in methanol.
Conclusions and questions:
People with access to zinc (not zinc oxide or any other form! has to be pure zinc) can reduce their own harmaline into THH easily at home. People need to be careful with hydrogen gas build up (we dont want explosions please!), so let the container vent and work in well ventilated area away from fire sources. There are still a couple of questions to be answered:
- Can we find other ways to separate THH from zinc carbonate without the need for solvents?
- Does 96% ethanol dissolve zinc carbonate in any significant quantity vs what it dissolves THH?
- Can zinc reduce harmine to harmaline too?
- What is the toxicity of zinc carbonate?
As always, im attaching the raw .MS file from agilent mass spec for those that want to double check my work, as well as the mass spectra of the detected THH peak
Pre-work:
Harmalas were extracted from syrian rue, precipitated mostly pure harmine (1 ) by adding sodium bicarbonate until no more color change was seen. This was left to stand for an hour, filtered to retrieve harmine, and then some sodium carbonate was added till no color change was noticed anymore, to precipitate a mixture of harmine/harmaline (2). This mixture (2) was filtered/dried and redissolved in acidic solution. To this solution, sodium bicarb was added to re-precipitate the harmine from mixture, which was filtered, and then sodium carb was added to precipitate reasonably pure harmaline (95%).
The reduction:
We dissolved the harmaline in some cheap supermarket vinegar, added excess zinc dust (which had been kept in a plastic bag for 2 years, we were affraid it had mostly oxidized and would not work). A lot of zinc dust just stayed on the bottom, and we could notice some very very fine bubbles in the vinegar (which is Hydrogen gas that is formed). We stirred occasionally and let it sit for a couple of hours. Finally, solution was filtered and sodium carbonate added. There was a lot of precipitation.
The precipitation was a mixture of alkaloids as well as zinc carbonate. In literature, this reduction is often done with HCl instead of vinegar, and then adding ammonium chloride, and later ammonium hydroxide to precipitate the base alkaloids. Apparently ammonium chloride prevents zinc salts from precipitating, so you only have your THH. But since we didnt really consider consuming and it was just a test, we precipitated with sodium carbonate and had excess zinc carbonate.
Zinc carbonate is NOT soluble in alcohols, so theoretically someone could use some ethanol to soak the alkaloid+zinccarbonate mixture and evap that to have alkaloids free of the zinc carb.. To analyze, after filtering this mixture, I soaked it in methanol, which was inserted in
The results:

GC-MS results show the conversion from harmaline to THH to be virtually 100%. There are no traces of harmaline detected, only small peak of harmine left (zinc is probably not strong enough to reduce harmine to harmaline but it might be it does, I cannot quantify that peak with the program I have it doesnt "detect" it).
There are other small peaks which are very likely the vinegar impurities (acetic compounds... one that seems to be glycerol acetate and other long chain compounds which seem to be carboxylic acids), Im not sure if any of these compounds were created from reduction of vinegar or if they are present in vinegar in the first place, but they are certainly not coming from the harmalas. There is one other small peak which is a phthalate, coming from plastic container used during dissolution of the alkaloids in methanol.
Conclusions and questions:
People with access to zinc (not zinc oxide or any other form! has to be pure zinc) can reduce their own harmaline into THH easily at home. People need to be careful with hydrogen gas build up (we dont want explosions please!), so let the container vent and work in well ventilated area away from fire sources. There are still a couple of questions to be answered:
- Can we find other ways to separate THH from zinc carbonate without the need for solvents?
- Does 96% ethanol dissolve zinc carbonate in any significant quantity vs what it dissolves THH?
- Can zinc reduce harmine to harmaline too?
- What is the toxicity of zinc carbonate?
As always, im attaching the raw .MS file from agilent mass spec for those that want to double check my work, as well as the mass spectra of the detected THH peak