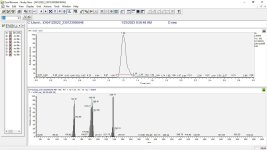
I recently did a run, extracted with aqueous NaOH and DCM, and ran the recrystallized product through the mass spec; my results were consistent with those in the attached paper (product 10); although I did extract quickly ( 0 mins), the exothermic conditions lead to boiling of the dcm in the sep funnel, and of course, heat catalyzes adduct formation. M.P. is 63-66C; I will experiment on recrystallizing under vacuum, maybe short-path, to eliminate the n-chloromethyl adduct. Another option would be to just salt it.
0 mins), the exothermic conditions lead to boiling of the dcm in the sep funnel, and of course, heat catalyzes adduct formation. M.P. is 63-66C; I will experiment on recrystallizing under vacuum, maybe short-path, to eliminate the n-chloromethyl adduct. Another option would be to just salt it.
-
Members of the previous forum can retrieve their temporary password here, (login and check your PM).
Reaction of N,N‑Dimethyltryptamine with Dichloromethane Under Common Experimental Conditions
- Thread starter benzyme
- Start date