This is what I call Vitamin P (see attachment below). It is a loose sparkly red powder composed of vitamin C, psilocin ascorbate, mushroom sugars that are not soluble in acetone (such as mannitol if present), and possibly other mushroom material.
I bioassayed 500mg of the powder by dissolving it in water (equivalent to 1.5g of dry mushroom) 18 days after preparing it, and it was active. Come on was quick. Can't tell what the % yield was, but it seemed reasonable based on the subjective bioassay.
Procedure
- 6g of pre-sporulating dried P. Cubensis fruits we're ground and placed in a mason jar.
- 500ml of acidic water (1g of vitamin C) we're added to the jar (pH ~ 4).
- After an hour rest, jar contents we're brought up to 70C in a hot water bath and allowed to cool (done 2x). In this step, psilocybin should be converted to psilocin (at this point we have been following this, except that Vitamin C is being used over vinegar to prevent oxidation.)
- Acid soup was filtered until only very slight cloudiness remained and it easily passed trough a triple stack of coffee filters.
- Acid was dried in a shallow pan in the oven at lowest setting.
- Resulting hard waxy film should contain psilocin ascorbate along with excess Vitamin C and other fungal material soluble in acidic water.
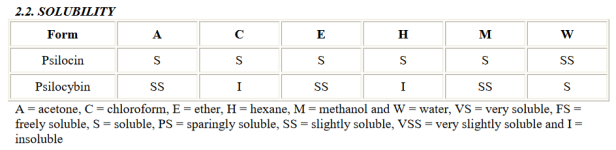
- Waxy material was washed with dry acetone twice (acetone washes are also used here) making sure all waxy flakes dissolve into the acetone and recovering the settlement with a coffee filter between washes.
- Dry weight of final material is 2g. 1g of this is mushroom material and 1g is vitamin C. The rest should be psilocin ascorbate (~100mg) which is (hopefully) a stable salt to oxidation when stored dry with Vitamin C
Vitamin P:
I bioassayed 500mg of the powder by dissolving it in water (equivalent to 1.5g of dry mushroom) 18 days after preparing it, and it was active. Come on was quick. Can't tell what the % yield was, but it seemed reasonable based on the subjective bioassay.
Procedure
- 6g of pre-sporulating dried P. Cubensis fruits we're ground and placed in a mason jar.
- 500ml of acidic water (1g of vitamin C) we're added to the jar (pH ~ 4).
- After an hour rest, jar contents we're brought up to 70C in a hot water bath and allowed to cool (done 2x). In this step, psilocybin should be converted to psilocin (at this point we have been following this, except that Vitamin C is being used over vinegar to prevent oxidation.)
- Acid soup was filtered until only very slight cloudiness remained and it easily passed trough a triple stack of coffee filters.
- Acid was dried in a shallow pan in the oven at lowest setting.
- Resulting hard waxy film should contain psilocin ascorbate along with excess Vitamin C and other fungal material soluble in acidic water.
- Waxy material was washed with dry acetone twice (acetone washes are also used here) making sure all waxy flakes dissolve into the acetone and recovering the settlement with a coffee filter between washes.
- Dry weight of final material is 2g. 1g of this is mushroom material and 1g is vitamin C. The rest should be psilocin ascorbate (~100mg) which is (hopefully) a stable salt to oxidation when stored dry with Vitamin C
Vitamin P:









 .
.