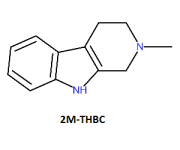
this betacarboline is associated with DMT containing species and is the betacarboline analog of DMT itself
One should be able to produce this chemical by employing a Pictet Spengler reaction on DMT, something typically requiring heat, a Lewis acid and aldehyde.
This betacarboline might be an artifact of hot acidic extractions, or perhaps it is present in the plant, however it might not be as soluble as DMT, typical exactions that result in traces of this alkaloid may not be ideal for it and may only be recovering a small portion of the molecule present in the basic or caustic solution.
This betacarboline may also oxidize.
It should have a glow under uv, unlike DMT, so if it did occur in DMT the material would glow in a visible spectrum under a black light. DMT is fluorescent, but not in a visible spectrum.
I would expect that this alkaloid, 2MTHbC, would also be colored, and would discolor DMT if it occurred in it.
I wish there was more data about central effects of this molecule. It occurs in nature in DMT containing plants and has been known as a contaminant product in DMT synthesis.
What are the sizes of the breakdown products of this molecule, what does it fracture into and how does it's molecular weight and melting point differ from DMT?
edited to add this :
2-Methyl-1,2,3,4-tetrahydro--carboline (4), white
shiny needle crystals (MeOH) (50 mg), mp 213 ∞C,
UV λmax (MeOH) nm 290, 294. EIMS, m/z (rel.
int.): [M+] 186 (26), 171 (6), 144 (100), 143 (63),
115 (10), 94 (5), 77 (4), 42 (4).