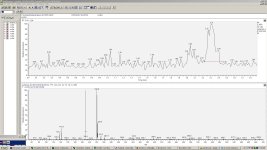
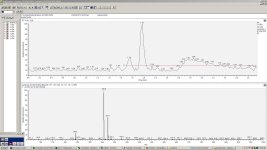
after much contemplation, I finally got around to prepping PFtek jars with 15 mM MiPT (made from tryptamine, derived from tryptophan) as an experimental group, and a control group without MiPT. Characterization will be by HPLC-UV-MS.
-
Members of the previous forum can retrieve their temporary password here, (login and check your PM).
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Biotransformation of MiPT to 4-HO-MiPT
- Thread starter benzyme
- Start date
Migrated topic.
Would this work as well with organisms that produce bufotenin?
downwardsfromzero
Boundary condition
Maybe not. The 5-hydroxylation of tryptophan occurs before decarboxylation in that pathway, I think. Not much is known about the isopropyl analogue of bufotenine.dragonrider said:Would this work as well with organisms that produce bufotenin?

Thanks for the support. It's been discussed before, I'm just fortunate of having the means of characterizing the end products. LCMS determination of psilocin/psilocybin in mushrooms is something I'm no stranger to, and I have spectra for the MiPT.
welp, those spores were nonviable. I just innoc'd some jars with a new syringe (most vendors have opted for syringes vs. prints), with 20 mM MiPT, and a control jar.
I also ordered a syringe, and a print, of c.cyanescens. That species may particularly have stronger expression of PsiH, a gene which encodes for tryptamine-4-monooxygenase (a CYP450 enzyme, nonspecific oxidoreductase). Said gene was only recently characterized, by Fricke et. al, 2017.
I also ordered a syringe, and a print, of c.cyanescens. That species may particularly have stronger expression of PsiH, a gene which encodes for tryptamine-4-monooxygenase (a CYP450 enzyme, nonspecific oxidoreductase). Said gene was only recently characterized, by Fricke et. al, 2017.
RoundAbout
Rising Star
I guess the main peak in the control is a fragment from the alpha cleavage of psilocybin, but what are the main peaks in the experimental? Out of curiousity...
RoundAbout said:I guess the main peak in the control is a fragment from the alpha cleavage of psilocybin, but what are the main peaks in the experimental? Out of curiousity...
your guess is as good as mine. I didn't run any standards