PsilocybeChild
Rising Star

Marine Indole Alkaloids
Marine indole alkaloids comprise a large and steadily growing group of secondary metabolites. Their diverse biological activities make many compounds of this class attractive starting points for pharmaceutical development. Several marine-derived indoles were found to possess cytotoxic...
Lot of interesting indole alkaloids here, some tryptamines, found in marine algaes and sponges.
For instance:
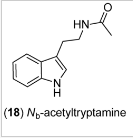
Psychedelic tryptamine?Examination of the organic extract of the broth of an unidentified algicolous fungus collected from the surface of the marine red alga Gracilaria verrucosa led to the isolation of Nb-acetyltryptamine
You can get this algae powdered on eBay under the listing title:
Aga Agar powder HOANG YEN - Gracilaria Verrucosa - Nutrient Agar
sold as a food ingredient.
Listing says "it's very suitable for people who are losing weight". 5-HTP (5-hydroxytryptamine) is an appetite suppressant. Nb-acetyltryptamine than could be responsible for their claimed weight-loss, suggesting perhaps their is active levels in the algae.