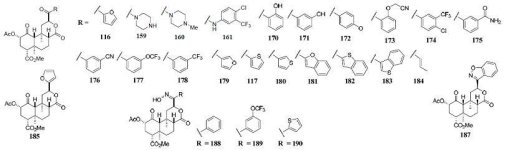
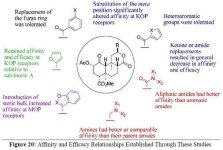
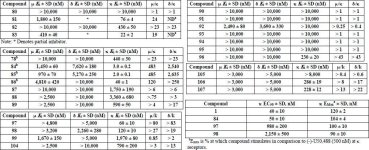
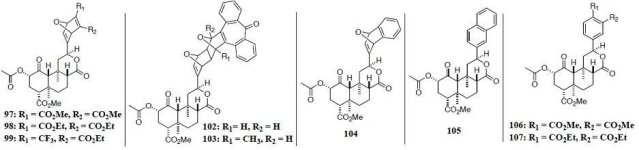
Here's some notes on another fairly technical dissertation that examines the biosynthetic routes in
S. divinorum that form salvinorin A and its neoclerodane brethren. [Since we don't have BB code for superscript text, all references to carbon-13 will simply be written as "13C"]
Kutrzeba, Lukasz. 2009. “Biosynthesis of salvinorin A, a potent hallucinogen from Salvia divinorum Epling & Jativa.” Ph.D. dissertation, Pharmaceutical Sciences: University of Mississippi.
First, a note on extraction of salvinorin A and related compounds: 10 kg fresh leaf were dipped in 1 liter of chloroform for 1 minute, yielding 8.54 g crude extract from which 2.42 g salvinorin A was purified by crystallization from ethanol (0.024% yield fresh weight or 0.2% yield dry weight), indicating efficiency comparable with much more laborious extractions previously reported in the literature. [Incidentally this indicates that dried leaves weigh 12% of the fresh mass, or that 1 g dried leaf is equivalent to 8.25 g fresh leaf]
Terpenoids are generally constructed from one of two pathways, both of which produce isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP). The
mevalonic acid pathway (MVA), also known as the HMG-CoA reductase pathway, was discovered first. The other route is the
2-C-methyl-D-erythritol 4-phosphate/1-deoxy-D-xylulose 5-phosphate pathway (MEP/DOXP), sometimes known as the non-mevalonate pathway. Archaea, animals, and fungi utilize the MVA pathway; green algae utilize the MEP pathway; some plants and bacteria are capable of using either.
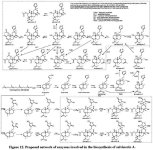
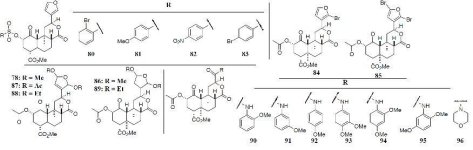
The proposed biosynthesis route from geranylgeranyl diphosphate is shown below:
(The poor readability of small text is due to the fact that a physical copy of the dissertation was submitted, rather than a pdf)
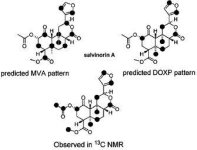
Kutrzeba examined the incorporation of 13C in salvinorin A by 13C NMR after feeding with 13C labeled glucose ([1-13C]glucose). Incorporation was entirely consistent with the MEP pathway. Incorporation of 13C in the C-23 methyl ester was speculated to be a result of methylation by enzymatic action of a type III S-adenosyl-L-methionine dependent O-methyltransferase. Labeling present in the acetate ester indicates that the labeled glucose also resulted in labeling of acetyl-CoA:
Experiments using uniformly labeled glucose ([U-13C]glucose) and [1-13C,2,4-2H]deoxy-D-xylulose similarly confirmed incorporation patterns consistent with the MEP pathway. Experiments with [Me-13C]L-methionine were limited due to toxicity, but confirmed that the labeled methyl group incorporated into C-23 in salvinorin A (consistent with the comment on type III SAM-dependent O-methyltransferases above).
Based on isolated genes for the peltate glandular trichomes (the structure to which salvinorin A is localized), Kutrzeba postulates that the terpenoid products of
S. divinorum are produced in those structures. He optimized a method for collecting the glandular trichomes by brief abrasion of young leaves with glass beads followed by straining through a series of meshes. He then isolated mRNA from the glands to produce cDNA, which was then sequenced. This turned up the presence of “one carboxy methyltransferase, five acetyl transferases, one diterpene synthase, six dehydrogenases, six reductases, and nine cytochrome P450’s.” At least three acetyltransferases are required to form S. divinorum neoclerodanes (one for C-1 acetylation in salvinorins C, D, and G; one for C-2 acetylation in salvinorins A, C, and E; one for C-17 acetylation in divinatorins C and D), though four or five could be needed depending on how specific the acetyltranferases are for their substrates. Dehydrogenases are required for the formation of the furan ring, for reduction of secondary alcohols to ketones (C-1 in salvinorins A and B, C-2 in salvinorin G), and for reduction of the C-17 alcohol to an aldehyde in divinatorin E; they may also be involved in formation of the lactone ring, though a CYP450 enzyme could similarly mediate that step. A reductase is likely involved in the reduction of the C-3/C-4 double-bond. CYP450 enzymes are involved in the formation of the C-18 acid, oxidation at C-1 and C-2 to form α-oriented hydroxyl moieties, oxygenation at C-12, possibly oxygenation at C-17, and possibly in the formation of the furan ring. The roles of the specific sequenced enzymes were assessed by comparison with known enzymes that have been sequenced and characterized from other plants.
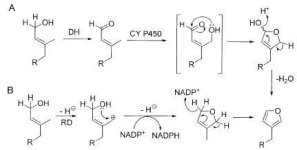
Two possible reaction schemes are proposed for the formation of the furan ring. Kutrzeba suggests that in either case this furan ring formation is likely the first step in the transformation from kolavenol (a simple clerodane structure formed from cyclization of geranylgeranyl diphosphate by a diterpene synthase) to the salvinorin and divinatorin compounds.
Kutrzeba suggests that since the C18 group exists as a free carboxylic acid in only 25% of the terpenoids isolated from S. divinorum (and as the methyl ester in the remainder, including in salvinorin A), we may suppose that methylation via a carboxy methyltransferase occurs early in the biosynthesis.
The carboxy methyltransferase found in the glandular trichomes was isolated and its activity characterized; it was found to be capable of methylating divinatorins A-C and hardwickiic acid, but not salvinorin A acid or salvinorin B acid, again supporting the notion that this methylation occurs early in the biosynthetic pathway.
I was going to refrain from critiquing errors in the paper, but some of them are bothersome enough that I feel obliged to point them out:
“With such exceptions as aminitin, hallucinogenic substances are generally considered physiologically safe.” Aminitin is not a hallucinogenic compound, it’s a toxin that occurs in some species in the genus
Amanita (as well as in other genera)… but not in
A. muscaria or
A. pantherina. The statement is nonsensical. It’s like saying: “With such exceptions as atropine, mashed potatoes are not generally considered hallucinogenic.”
Kutrzeba talks too often of proof: about an experiment “proving the involvement of KOR in modulation of mood states” or that “models proved the hypothesis…” While his sentence construction indicates that English is not his first language, as a scientist he should know that the word “proof” should be assiduously avoided in any but the most extreme circumstances. Data and experiments can support a hypothesis or model, but claiming to prove the hypothesis implies that you know the outcome of every single future experiment that will ever be performed.
Like many others, Kutrzeba cites Valdés
et al. 1983 as the source for the claim that
S. divinorum has been used for centuries. Valdés
et al. note that the use of mushrooms and morning glory seeds were discovered centuries ago, and that all literature on
S. divinorum is much more recent. They also reject its identification as
pipiltzintzintli and make no claims whatsoever on the antiquity of Mazatec use of the plant. I really don't understand why everyone seems to cite this paper on the subject of the antiquity of
S. divinorum use.