Samvidbuho
Esteemed member
So this is cool.. Thought I'd share a novel method of extraction using a super wicked acoustic technology. I too am excited because this is the very first application of using resonant acoustic technology for means of extraction. It represents one of the most efficient and fastest extracting technology out there although I cannot provide the evidence for this yet. Undoubtedly this use will surface, but for here, now, let me be the first to demonstrate:
I have in my lab a mixer called a Resonant Acoustic Mixer, or RAM for short. Applications are vast, from mixing of pharmaceutical powders, to mixing of concrete, but also its uses encompass ASD creation through acoustic fusion, nanoscale particle size reduction, kind of you name it.
Obviously, when I first heard about it I thought of extraction scenarios, and now that I have one to play around with I thought I'd test out the waters.
Oh by the way, it oscillates up and down at 60 times per second (resonant frequency ~60Hz) and up to 100g of acceleration as its moving. Think of it as 60fps at 980 m/s per second.
I did 3 things:
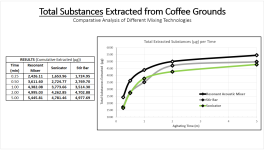
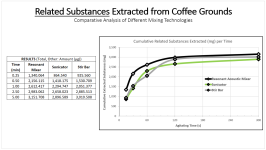
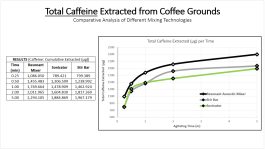
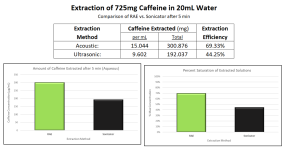
1. Demonstrated the use of Resonant Acoustic Mixing for extraction purposes. You have your typical extraction brew (solids filtered out), basic pH, and on top you can have your solvent, here toluene. The collage below represents 8 seconds of acoustic mixing at 100g. The photos say it best, but what's happening is "Faraday instabilities" are being created through the oscillating action, which leads to finger-like projects extending upwards and downwards like stalactites and stalagmites. The surface area of the liquids is amplified to a crazy degree, and micromixing occurs in the bulk. All energy is directed towards the brew and a computer automatically monitors the power and adjusts for the constant resonance.
I did not have anything left to extract in this case so I don't have color changes in solvent to show, but it's suitable enough to show what could be possible
2. I thought maybe I could mix 2 immiscible liquids, methanol and heptane. I used a small bit of methanol to solibulize some spice and added heptane on top. I hoped that the mixing would transfer some of the solids to the heptane. The collage in this case shows 30 seconds of mixing with complete binding into a single phase of solution.
3. Supersaturation: Last scenario is I have some crystals contained with an amount of heptane. Here I hoped that the mixing would be so efficient that supersaturation would happen, and crystallization would happen without any heating. There was particle size reduction but not any supersaturation after 30 seconds. Mixing for longer could definitely make a difference, though. For obvious reasons there is concern when mixing any solvent that's heated, but there are workarounds for that that could be explored further.
Nothing much to report on the visuals before and after, but I have a super slow-mo video that I took of the crystals being mixed.
Just wait until the slow-mo stops
Hope everybody finds this as cool as I do !
I have in my lab a mixer called a Resonant Acoustic Mixer, or RAM for short. Applications are vast, from mixing of pharmaceutical powders, to mixing of concrete, but also its uses encompass ASD creation through acoustic fusion, nanoscale particle size reduction, kind of you name it.
Obviously, when I first heard about it I thought of extraction scenarios, and now that I have one to play around with I thought I'd test out the waters.
Oh by the way, it oscillates up and down at 60 times per second (resonant frequency ~60Hz) and up to 100g of acceleration as its moving. Think of it as 60fps at 980 m/s per second.
I did 3 things:
1. Demonstrated the use of Resonant Acoustic Mixing for extraction purposes. You have your typical extraction brew (solids filtered out), basic pH, and on top you can have your solvent, here toluene. The collage below represents 8 seconds of acoustic mixing at 100g. The photos say it best, but what's happening is "Faraday instabilities" are being created through the oscillating action, which leads to finger-like projects extending upwards and downwards like stalactites and stalagmites. The surface area of the liquids is amplified to a crazy degree, and micromixing occurs in the bulk. All energy is directed towards the brew and a computer automatically monitors the power and adjusts for the constant resonance.
I did not have anything left to extract in this case so I don't have color changes in solvent to show, but it's suitable enough to show what could be possible
2. I thought maybe I could mix 2 immiscible liquids, methanol and heptane. I used a small bit of methanol to solibulize some spice and added heptane on top. I hoped that the mixing would transfer some of the solids to the heptane. The collage in this case shows 30 seconds of mixing with complete binding into a single phase of solution.
3. Supersaturation: Last scenario is I have some crystals contained with an amount of heptane. Here I hoped that the mixing would be so efficient that supersaturation would happen, and crystallization would happen without any heating. There was particle size reduction but not any supersaturation after 30 seconds. Mixing for longer could definitely make a difference, though. For obvious reasons there is concern when mixing any solvent that's heated, but there are workarounds for that that could be explored further.
Nothing much to report on the visuals before and after, but I have a super slow-mo video that I took of the crystals being mixed.
Just wait until the slow-mo stops

Hope everybody finds this as cool as I do !