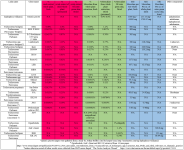
Adducts are common in LC-MS. Use attached table to help look for potential adducts.
Looking at the pdf's of the unknown peaks I had one clarification. Is the MS2 spectrum a secondary MS of the chosen ion? I assume they doing MS/MS here and that is why there are more then 1 spectra per peak?
After extracting T. Pachoni with acid (acetic acid 5%) / base (NaOH pH ~12.6) -> partition with chloroform few times to extract freebase alkaloids and salt with HCl acetone / IPA clean up ended up with material that tested by GC-MS contained:
Mescaline as major alkaloid
in addition to minor compounds:
3,4-dimethyoxy-phenylethylamine
1,2,3,4-tetrahydroisoquinoline-6,7,8-trimethoxy-1-methyl
anhalinine
If the chloroform layer was analyzed after removing the vast majority of mescaline without making salts just leaving as freebase there were more potential alkaloids left over:
anhalonine
carnegine
anhalinine
anhalidine
anhalonidine
Unfortunately the original mass spec data was lost when a computer and hard drive was stolen :x
Looking at the pdf's of the unknown peaks I had one clarification. Is the MS2 spectrum a secondary MS of the chosen ion? I assume they doing MS/MS here and that is why there are more then 1 spectra per peak?
After extracting T. Pachoni with acid (acetic acid 5%) / base (NaOH pH ~12.6) -> partition with chloroform few times to extract freebase alkaloids and salt with HCl acetone / IPA clean up ended up with material that tested by GC-MS contained:
Mescaline as major alkaloid
in addition to minor compounds:
3,4-dimethyoxy-phenylethylamine
1,2,3,4-tetrahydroisoquinoline-6,7,8-trimethoxy-1-methyl
anhalinine
If the chloroform layer was analyzed after removing the vast majority of mescaline without making salts just leaving as freebase there were more potential alkaloids left over:
anhalonine
carnegine
anhalinine
anhalidine
anhalonidine
Unfortunately the original mass spec data was lost when a computer and hard drive was stolen :x