Taken From this thread, post #75:
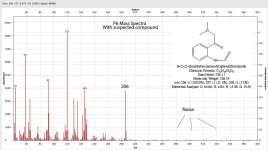
endlessness said:Here's the mass spectra file of the N-Oxide analysis I tried to do (10% hydrogen peroxide mixed to DMT in IPA)
Regarding N-Oxides, they do appear in GC-MS, except part of it seems to convert back to the parent compound. In this paper, they say:
approximately one third of clozapine-N-oxide was determined to be reduced to clozapine under the described capillary GC conditions. On-column reductions of N-oxides to the corresponding tertiary amines find a precedent in the property of other tricyclic drug N-oxides subjected to the generally more reactive conditions of packed column GC [12-14]. (...) thermal degradation of this Noxide to the parent drug may result in an overestimation of clozapine concentrations
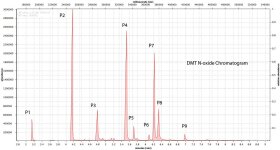
Now interestingly, when i tried to make an n-oxide with 10% hydrogen peroxide, it did seem to create the dmt n-oxide peak (eluted at around 5.7, with the mass peak at 204, in the file im attaching, same as with FASW file eluted at around 5.72). But if we take the above paper to be right also for DMT, then the n-oxide amount is probably being under-estimated because some of it is converting back to the parent compound. LC-MS will help settling this. The other interesting thing is that the supposed N-Oxide product I created has several other peaks appart from dmt n-oxide and DMT.. It also has a very large quantity of 2MTHBC and several other components.. In the paper mentioned above, when analyzing the pure n-oxide, they also came up with 3 peaks instead of one.. Here's what they said:
Formation of these latter components may represent thermally induced Meisenhiemer rearrangement [15] (C-N oxygen insertion) and/or enamine forming processes [13,14], both characteristic GC degradation pathways of N-oxides.
I wonder how much of this explanation is valid for the other compounds we're seeing in the n-oxide analysis, and how much are actually other degradation products of DMT .. ? Even in the TLC of the n-oxide there were other peaks appearing, so maybe its not only in the gc-ms but DMT can oxidize to other things with hydrogen peroxide, it seems (maybe also with air.. ?) Here's the TLC Plate:

(made with adding 20mg dmt to a petri dish, adding a few ml hydrogen peroxide to see some of it dissolve, then add some IPA and more hydrogen peroxide for it all to dissolve
Also compare it with last column in TLC plate in the first post of this thread
Maybe 3 = 2MTHBC ?
? Now take into account what infundibulum said to me by pm (hope you dont mind me sharing, inf) :
This is basic rule in chemistry and oxidation is such a generic term and used to describe an array of diverse reaction and products. Believe it or not, the cyclisation of dmt to 2MTHBC is actually an oxidation reaction!
Burning dmt is also an oxidation reaction, an extreme one; all organic substances burn (or oxidise) to CO2 and H2O. So an oxidation product of dmt is carbon dioxide and water, also some nitrogenous compounds due to the presence of nitrogen as well. But there are so many other oxidation intermediates and by-products, like 2MTHBC as mentioned
I also googled "indole oxidation" just to get a rough idea of what else can happen to dmt and damn, it is diverse! one can get indole fusion by products (like in here: http://www2.iq.usp.br/docente/lhc/lab/indole.pdf) or just addition of oxygen atoms such as here: http://en.wikipedia.org/...dole_NBS_Oxidation.png. And psilocin also is thought to be oxidising to a blue compound, maybe of similar structure to indigo (File:Indigo.svg - Wikipedia).
So when one employs H2O2 he's sure to oxidise the tertiary amine of dmt to n-oxide, but he may be oxidising the molecule in who knows in how many other different ways. We may also need to take into account that some oxidations may be fast (like the oxidation of n to n-oxide) but others may be slower. Once I tried to make some n-oxide using peracetic acid and yes, it turned deep red pretty quickly (maybe n-oxide?). I left it alone for few days and the oxidation appeared to be proceeding and the deep red started changing to clearer yellow (maybe hell knows further oxidised products?)
And with air oxidation, who really knows what may be happening; some n-oxide formation? some other oxidation by products? Still all too interesting and crying to be tested!
How interesting, that 2mthbc amount is increasing a lot when doing this oxidation test with hydrogen peroxide, as inf said, its also an oxidation reaction.... But wow, can it really be?