(edit: New analysis from 2023)
Mimosa hostilis rootbark (#148 )
Jan 2023

Origin: : Brazil
Results:
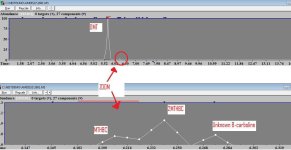
DMT 0.85%
NMT 0.52%
Yuremamine 0.35%
DMT N-oxide 0.05%
Bufotenine 0.003%
Mimosa hostilis rootbark (#149)
Jan 2023

Origin: : Mexico
Results:
DMT 0.76%
NMT 0.35%
Yuremamine 0.29%
DMT N-Oxide 0.005%
Bufotenine 0.003%
Mimosa hostilis (#150)
Jan 2023

Origin: : Mexico
Results:
DMT 0.71%
NMT 0.45%
Yuremamine 0.13%
DMT N-Oxide 0.005%
2-MTHBC 0.002%
Bufotenine 0.001%
Other excellent analysis from Brennendes Wasser:
Older analysis from 2012 below:
Mimosa tenuiflora (syn. hostilis)/Jungle Spice Analysis (last update oct 2012)
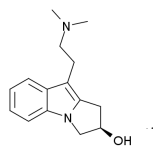
Mimosa tenuiflora is one of the most important DMT-containing plants. It is used traditionally in Northeast Brazil and has been sold for years by several ethnobotanical suppliers. For these reasons, it has been the focus of considerable research by Nexians. We have tested both the crude plant preparations as well as different products extracted using a variety of different methods.
Methodology
At first I started with TLC vs some reference standards, and this has eventually evolved to GC-MS and LC-MS testing of all samples
The main results are:
Orange text indicates novel research.
++++ indicates the main substance present
+++ indicates relatively high amounts present
++ indicates moderate amounts present
+ indicates small amounts present
- indicates trace amounts present
Mimosa tenuiflora (whole root bark):
++++ DMT
+++ Yuremamine
++ Catechol, homocatechol, resorcinol, etc. (possibly breakdown products of yuremamine and/or lignin) * (these have been tentatively identified by mass spectra but not confirmed with a reference standard)
+ NMT
+ 2-MTHBC
- Hordenine, N-Methyl-Phenetylamine, Dimethyl-Phenetylamine, N-Formyl-NMT, 1,2-Dimethyl-Tetrahydro-Beta-Carboline * (these last 5 trace alkaloids have been tentatively identified by mass spectra but not confirmed with a reference standard)
DMT (extracted with naphtha, xylene + FASA or limonene + FASI):
++++ DMT
+ NMT
+ 2-MTHBC
DMT (extracted with xylene + evaporated or limonene/xylene/toluene + aqueous
saltings):
++++DMT
+NMT
+2MTHBC
+ 1-2-Dimethyl-Tetrahydro-Beta-Carboline.
+ 1-Methyl-Tetrahydro-Beta-Carboline
- Hordenine, N-Methyl-Phenetylamine, Dimethyl-Phenetylamine, N-Formyl-NMT * (these last 6 trace alkaloids have been tentatively identified by mass spectra but not confirmed with a reference standard)
Conclusions.
These analyses confirmed that heating up or extracting Mimosa tenuiflora (and analysis by GC-MS) breaks down some of the original components found in the plant material, most notably yuremamine, which may play a role in making cold-brewed Mimosa preparations orally active without a RIMA/MAOI (Callaway 2005)
When extracting with solvents that are less-selective than naphtha, we found very small amounts of two other beta carbolines that have unknown effects. It's possible that at this concentration they do not affect the experience, but they might, at these or higher concentrations.
Some people believe that even in the small amounts found, these alkaloids do
affect the experience but we don’t yet know if this is the result of self-suggestion or not. The few psychonautic blind tests that have been conducted do not confirm any noticeable difference in activity.
To concentrate these non-DMT alkaloids, you can precipitate DMT from a solvent by the FASA/FASI method, and later wash the solvent with water and evaporate the water to retrieve the other alkaloids plus excess fumaric acid and some other plant impurities.
It is important to note that we do not know if a concentration of these other alkaloids is safe or desirable for ingestion. We also have yet to confirm whether at least part of these other alkaloids are artifacts of extraction/analysis or if they are present in the plant. Identification of main substances was done with reference standards and GC/LC-MS but some of the more obscure substances present were only based on mass spectra match in database after GC-MS testing, so should not be considered an absolute proof. I post further in this thread and elsewhere in this subforum the attached mass spectra and welcome any and all peer review.
Original post from dec 2011
Here's some more analysis ive been doing lately on extracts of Mimosa Hostilis
This is all from A/B extraction on Mimosa hostilis using lye as a base (and limonene as solvents in most of them, except 6 and 9, which are using naphtha, and 5, using xylene)
First, the samples:
J - Jungle - Xylene evap and then 2x warm naphtha pulls to supposedly remove (most) DMT
1- Jungle - Xylene evap
2- FASW (Fumaric Acid Saturated Water) crude saltings from limonene pulls
3- FASI (Fumaric Acid Saturated IPA) precipitation from limonene pulls - From pulls 2,3,4, yellow
4- FASI - From pull 1, white
5- FASA (Fumaric Acid Saturated Acetone) precipitation from xylene pulls.
6- FASI - Yellow part on the bottom of FASI crystallization, due to water in IPA
7- Freebase from naphtha, white
8- Freebase from converted FASI, white
9- Freebase from naphtha, yellow.
10- Freebase from converted FASW, brown impure
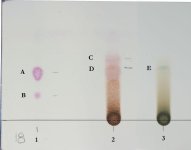
Now TLC plate:

The last two columns: Second last is some attempt to make n-oxide (just added 10% hydrogen peroxide to a bit of sample 7 dissolved in methanol and used that. I think 10% is too weak to make full conversion but clearly there was some change). Last column is DMT standard.
As you can see, samples 2,3,4,5 and 6 all have a top streak/spot, which is the fumaric acid. Sample J has some spots/streak at around same height but its clearly some different substance/s.
Most samples, except 8, have DMT and NMT spots. Sample 8 was not done from same mimosa, was from earlier batch, so hard to make conclusions regarding apparent lack of NMT. (see later GC-MS discussion for more mistery... )
Samples J, 1, 2 and 6 show some Beta carboline, others dont see to show it (again, GC-MS will show more weirdness, stay tuned)
The first funny things that show up in this plate: the supposed no-dmt jungle seems to have plenty of dmt. Also the NMT presence in nearly all samples is pretty interesting. More information below with GC-MS analysis (and NMR soon)
And for those that care: This was done on silica gel 60 F254 from Merck, eluent is Methanol:Ammonium Hydroxide 100:2.5
The measured Rfs are:
DMT - 0.4-0.44
NMT - 0.19-0.23
Bcarboline - 0.03
N-oxide (?) - 0.31-0.34
Next up: GC-MS of samples 1,2,4,6,7 , and NMR of samples J, 1 and 2
Mimosa hostilis rootbark (#148 )
Jan 2023

Origin: : Brazil
Results:
DMT 0.85%
NMT 0.52%
Yuremamine 0.35%
DMT N-oxide 0.05%
Bufotenine 0.003%
Mimosa hostilis rootbark (#149)
Jan 2023

Origin: : Mexico
Results:
DMT 0.76%
NMT 0.35%
Yuremamine 0.29%
DMT N-Oxide 0.005%
Bufotenine 0.003%
Mimosa hostilis (#150)
Jan 2023

Origin: : Mexico
Results:
DMT 0.71%
NMT 0.45%
Yuremamine 0.13%
DMT N-Oxide 0.005%
2-MTHBC 0.002%
Bufotenine 0.001%
Other excellent analysis from Brennendes Wasser:
Older analysis from 2012 below:
Mimosa tenuiflora (syn. hostilis)/Jungle Spice Analysis (last update oct 2012)
Mimosa tenuiflora is one of the most important DMT-containing plants. It is used traditionally in Northeast Brazil and has been sold for years by several ethnobotanical suppliers. For these reasons, it has been the focus of considerable research by Nexians. We have tested both the crude plant preparations as well as different products extracted using a variety of different methods.
Methodology
At first I started with TLC vs some reference standards, and this has eventually evolved to GC-MS and LC-MS testing of all samples
The main results are:
Orange text indicates novel research.
++++ indicates the main substance present
+++ indicates relatively high amounts present
++ indicates moderate amounts present
+ indicates small amounts present
- indicates trace amounts present
Mimosa tenuiflora (whole root bark):
++++ DMT
+++ Yuremamine
++ Catechol, homocatechol, resorcinol, etc. (possibly breakdown products of yuremamine and/or lignin) * (these have been tentatively identified by mass spectra but not confirmed with a reference standard)
+ NMT
+ 2-MTHBC
- Hordenine, N-Methyl-Phenetylamine, Dimethyl-Phenetylamine, N-Formyl-NMT, 1,2-Dimethyl-Tetrahydro-Beta-Carboline * (these last 5 trace alkaloids have been tentatively identified by mass spectra but not confirmed with a reference standard)
DMT (extracted with naphtha, xylene + FASA or limonene + FASI):
++++ DMT
+ NMT
+ 2-MTHBC
DMT (extracted with xylene + evaporated or limonene/xylene/toluene + aqueous
saltings):
++++DMT
+NMT
+2MTHBC
+ 1-2-Dimethyl-Tetrahydro-Beta-Carboline.
+ 1-Methyl-Tetrahydro-Beta-Carboline
- Hordenine, N-Methyl-Phenetylamine, Dimethyl-Phenetylamine, N-Formyl-NMT * (these last 6 trace alkaloids have been tentatively identified by mass spectra but not confirmed with a reference standard)
Conclusions.
These analyses confirmed that heating up or extracting Mimosa tenuiflora (and analysis by GC-MS) breaks down some of the original components found in the plant material, most notably yuremamine, which may play a role in making cold-brewed Mimosa preparations orally active without a RIMA/MAOI (Callaway 2005)
When extracting with solvents that are less-selective than naphtha, we found very small amounts of two other beta carbolines that have unknown effects. It's possible that at this concentration they do not affect the experience, but they might, at these or higher concentrations.
Some people believe that even in the small amounts found, these alkaloids do
affect the experience but we don’t yet know if this is the result of self-suggestion or not. The few psychonautic blind tests that have been conducted do not confirm any noticeable difference in activity.
To concentrate these non-DMT alkaloids, you can precipitate DMT from a solvent by the FASA/FASI method, and later wash the solvent with water and evaporate the water to retrieve the other alkaloids plus excess fumaric acid and some other plant impurities.
It is important to note that we do not know if a concentration of these other alkaloids is safe or desirable for ingestion. We also have yet to confirm whether at least part of these other alkaloids are artifacts of extraction/analysis or if they are present in the plant. Identification of main substances was done with reference standards and GC/LC-MS but some of the more obscure substances present were only based on mass spectra match in database after GC-MS testing, so should not be considered an absolute proof. I post further in this thread and elsewhere in this subforum the attached mass spectra and welcome any and all peer review.
Original post from dec 2011
Here's some more analysis ive been doing lately on extracts of Mimosa Hostilis
This is all from A/B extraction on Mimosa hostilis using lye as a base (and limonene as solvents in most of them, except 6 and 9, which are using naphtha, and 5, using xylene)
First, the samples:
J - Jungle - Xylene evap and then 2x warm naphtha pulls to supposedly remove (most) DMT
1- Jungle - Xylene evap
2- FASW (Fumaric Acid Saturated Water) crude saltings from limonene pulls
3- FASI (Fumaric Acid Saturated IPA) precipitation from limonene pulls - From pulls 2,3,4, yellow
4- FASI - From pull 1, white
5- FASA (Fumaric Acid Saturated Acetone) precipitation from xylene pulls.
6- FASI - Yellow part on the bottom of FASI crystallization, due to water in IPA
7- Freebase from naphtha, white
8- Freebase from converted FASI, white
9- Freebase from naphtha, yellow.
10- Freebase from converted FASW, brown impure
Now TLC plate:

The last two columns: Second last is some attempt to make n-oxide (just added 10% hydrogen peroxide to a bit of sample 7 dissolved in methanol and used that. I think 10% is too weak to make full conversion but clearly there was some change). Last column is DMT standard.
As you can see, samples 2,3,4,5 and 6 all have a top streak/spot, which is the fumaric acid. Sample J has some spots/streak at around same height but its clearly some different substance/s.
Most samples, except 8, have DMT and NMT spots. Sample 8 was not done from same mimosa, was from earlier batch, so hard to make conclusions regarding apparent lack of NMT. (see later GC-MS discussion for more mistery... )
Samples J, 1, 2 and 6 show some Beta carboline, others dont see to show it (again, GC-MS will show more weirdness, stay tuned)
The first funny things that show up in this plate: the supposed no-dmt jungle seems to have plenty of dmt. Also the NMT presence in nearly all samples is pretty interesting. More information below with GC-MS analysis (and NMR soon)
And for those that care: This was done on silica gel 60 F254 from Merck, eluent is Methanol:Ammonium Hydroxide 100:2.5
The measured Rfs are:
DMT - 0.4-0.44
NMT - 0.19-0.23
Bcarboline - 0.03
N-oxide (?) - 0.31-0.34
Next up: GC-MS of samples 1,2,4,6,7 , and NMR of samples J, 1 and 2