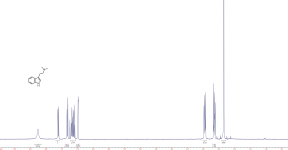
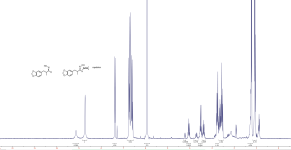
Cheelin said:
I am not denying that yellow cannot be dmt, and never have.
Some seem unable to admit that yellow can also be contaminated dmt.
Come on, get off the high horses, and down into the crystallization trenches, with some practical applications, rather than arguing about nonsense.
In other words, aren’t the goos more likely to simply be contamination in these circumstances?
You need to be specific. This is why you got in a spat with benzyme and got all defensive. What is "goos"? is goo supposed to mean just anything and everything that came out of a dmt extraction, no matter how sloppy it was carried out? Be specific. If it is goo that has some yellow black tar in it because you were sloppy in the extraction, then specify that. Then it becomes very easy to make the distinction where the impurity is coming from. Usually when people ask questions, its AFTER they followed everything that was neatly written for them in the cookbook tek. Otherwise I could have just as easily pissed on my crystal dmt and it would be equally yellow goo by your definition. So when someone posts 'what is my goo?' without being specific you have to basically assume that they could follow the tek and not spill any tar into it, because if they are asking the question why is my DMT goo you would hope they have at least covered the bases properly and not made any obvious mistakes that would account for discolouration or 'goo'.
Perhaps do the experiment, decant an amount of loaded solvent with a few drops of the tar…look at the pics of dmt that some people post, it is not all pure dmt that is yellow.
What a ridiculous argument that yellow product is necessarily pure dmt.
Again, really?
I guess i don’t see the purpose of theoretical discussions that don’t provide practical applications on a site like this. My bad.
Last time I checked, this thread was called Advanced/Enhanced chemistry » Polymorphic properties of DMT
not
Basic DMT Extraction help » Cheelin's extraction philosophy
Your 1-3 word posts are also bad mannered and not appreciated by anyone on the forum, since it forces people to click page after page to find the posts that actually have content.
I accept your statement about your intent. You comments (as well as others on this site, at your chemistry level), here and in other threads, do come across to lowlings like me, as patronizing and smug, sometimes. I have experience in a number of “expert” cohorts, and this is very common when dealing with “non-experts”…something to be mindful of.
As far as “tall order”, wrt my “Maybe” comment (which appears not to be completely understood): it may seem so, but i think not.
For example, i think, and the literature shows, there are much faster ways to extract alkaloids from roots and lignified plant tissue. Yes, much of it is beyond kitchen chemistry, but if I had the appropriate chemistry background, i bet I could adapt some of this technology for our circumstances. An easier to defend example is Loveall’s work (as well as his humble approach, given his obvious expertise): he has already demonstrated significant pull process time reduction vs Max Ion (while maintaining good yield and WHITE crystals).
If you don’t set high, but achievable goals, you keep using old technology. I look forward to seeing some of the brainpower and expertise on this site, get back in the game of improving the extraction technology that was developed going on 10 years ago.
have you ever heard the expression, give a man a fish, feed him for a day, teach a man to fish, feed him for a lifetime?
You've been here what, 4 months?
Its like your walking into a Michelin star restaurant, as a dishwasher on day 1. You ask the chef, "hey what do you think of my Bolognese? I tried this and that and it tasted like this." The chef responds not in the way you were expecting, instead of giving you a recipe for a Bolognese, he starts talking about things like flavor, fat and acidity balance, tasting the sauce and adjusting. And you hear that and begin to get frustrated "Why won't you give me a straight answer? why is he telling me all this esoteric nonsense why can't he just give me his recipe! He must be trying to guard it because hes smug!"
The problem is that you are looking for a new cookbook, the chef is trying to teach you how to think so you can write your own cookbook.
I listen to a lot of experts too in fields I am less familiar with, but I very rarely find them to be smug, very much the opposite. When I first joined this forum 12 years ago, benzyme was my senior and I didn't find him smug either. Actually the people who tend to be the most smug and conceited, are typically the ones who think they are experts but really don't know what they are talking about. They always eventually get found out, too. Loveall is a very smart and humble guy, but that is because he has an attitude which is the opposite of yours. He is always learning and he is always enthusiastic, he doesn't get defensive when he is wrong about something, he digs deeper so that he can understand it, he has good intuitions and knows his limitations, I admire him too.
Come on, get off the high horses, and down into the crystallization trenches, with some practical applications, rather than arguing about nonsense.
Again, really? I'm still really struggling to find justification for such poor attitude. Sounds more like its coming from someone with a bruised ego because they fell off their high horse and is eager to get back on. Read the part again about the Bolognese.
---