Here is something I found about TLC with Rf's for identifying mescaline which could be useful for the excel.
Maybe it's already posted somewhere here on the forum but I couldn't find it with the search.
It's coming from linked source which also has several other methods of mescaline identification described. According to the pdf it's referenced from
J. "Clarke's Isolation and Identification of Drugs"': 2nd edifion, The Pharmaceutical Press (London) 1986.
The Analysis of Peyote Cactus, Mescal Buttons and Mescaline
E. Thin-layer chromatography
Plates:
Activated silica gel G on glass backed plates; the coating (0.25 mm thickness) contains a fluorescing additive which fluoresces at 254 nm.
Developing solvents:
System A: Chloroform 82 Methanol 17
Concentrated ammonia solution 1 System B: Methanol 100
Concentrated ammonia solution 1.5 Preparation of solutions to be applied to the TLC plate:
a) Peyote cactus, Mescal Buttons:
About 10 mg of a representative sample of the suspected dried and pulverized
plant material is extracted using the method outlined in Chapter B. If using ninhydrin reagent for visualization the extract should be concentrated to approxymately 200 ul under a stream of nitrogen. and 10 ul of the filtered extract are spotted onto the TLC plate.

b) Mescaline powder:
Prepare a solution at a concentration of approximately 1 mg/ml methanol as
outlined in Chapter B. and spot 1 ul of this solution onto the TLC plate.
c) Mescaline tablets and capsules:
Extract the material using method outlined in Chapter B. and spot 10 ul of this
extract onto TLC plate. d) Standard solution:
Prepare a solution of 1 mg/ml mescaline sulfate or hydrochloride methanol and spot 1 ul of this standard solution onto the TLC plate.
Visualization
The plates must be dried prior to visualization. This can be done by air-drying at room temperature or by short use of a hot air blower.
Visualization methods:
1. UV light at 254 nm.
2. Fluorescamine (Fluram) reagent 3. Ninhydrin reagent.
Spray reagents:
(1) Fluorescamine reagent: Prepare a solution of 10 mg fluorescamine in 50ml
dried acetone.
(2) Ninhydrin reagent: Prepare a 10% solution of ninhydrin in ethanol.
Method:
Observe first the plate under UV light at 254 nm. Mescaline will absorb the light
and appear as a dark spot on the fluorescent background. Then spray with reagent(1) or (2). When using reagent (1), the plate has to be dried after spraying by shortly using a hot air blower. Then observe the plate under UV light at 365 nm.
Mescaline gives a bright yellow fluorescent spot. The detection limit is about 10 ng mescaline. The fluorescence can be intensified by exposing the plate to ammonia vapour. When using reagent (2) the plate has to be heated after spraying in an oven at 120°C for at least 15 minutes. A violet spot is given by mescaline. The detection limit is about 1 ug mescaline.
Results
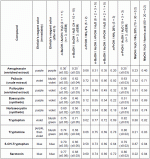
With developing systems A and B, mescaline gives spots at Rf x 100 value
approximating 36 and 24, respectively.