I also can't seem to find much info on the plants going to seed.
Are they sensitive to the light cycle or do they just go to seed after reaching maturity regardless?
My plants seem to be growing well and do show a fair amount of morphological differences already.
Any speculations on hybridizing this with P. aquatica or other P. species?
What morphologies should I look for? Obviously good spreading is important, but right now I'm mostly seeing tall, thin, weaker looking single blades and two samples producing more branching blades. Conditions haven't been quite identical yet, but hopefully I've got that worked out now.
The two lower germination rate samples seem to be producing more branching, so I'm wondering about that too now.
-FF
Are they sensitive to the light cycle or do they just go to seed after reaching maturity regardless?
My plants seem to be growing well and do show a fair amount of morphological differences already.
Any speculations on hybridizing this with P. aquatica or other P. species?
What morphologies should I look for? Obviously good spreading is important, but right now I'm mostly seeing tall, thin, weaker looking single blades and two samples producing more branching blades. Conditions haven't been quite identical yet, but hopefully I've got that worked out now.
The two lower germination rate samples seem to be producing more branching, so I'm wondering about that too now.
-FF


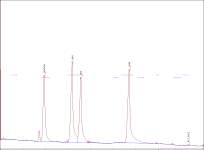
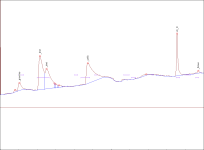
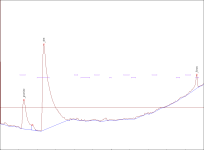
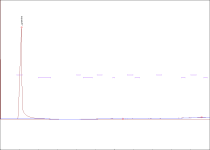
 ) and I took a look at this post and the thing that got my eye was Justin Case's report saying that after the grass is cut the enzymes transmute the NN into 5 MeO ... so what can you guys suggest me so I can stop it(It's not that fatal if I don't get N,N ,5MeO is still a nice tryptamine
) and I took a look at this post and the thing that got my eye was Justin Case's report saying that after the grass is cut the enzymes transmute the NN into 5 MeO ... so what can you guys suggest me so I can stop it(It's not that fatal if I don't get N,N ,5MeO is still a nice tryptamine  . The reagents will include citric acid(should I use vinegar because the one offered at my place is only 5% acetic acid),lye,and pure petroleum ether (which is Naphtha I believe).
. The reagents will include citric acid(should I use vinegar because the one offered at my place is only 5% acetic acid),lye,and pure petroleum ether (which is Naphtha I believe).