Summary: I grew six individuals each of Phalaris aquatica and P. brachystachys, indoors under lights. I extracted the leaves and analyzed them by HPLC. P. aquatica usually contained DMT, but 1/6 contained 5-MeO-DMT, consistently across two harvests. P. brachystachys always contained mostly 5-MeO-DMT, much less DMT, and significant amounts of other alkaloids, probably including but not limited to gramine.
Inspired by prior work on this forum and elsewhere, I've been experimenting with Phalaris, with an eventual goal of selecting or breeding for alkaloid content. My results aren't very useful yet, but I still wanted to share, especially since much of the literature on Phalaris predates modern analytical methods. This work predates my similar experiments with Sceletium (also posted in this forum), and this result is significantly less exciting to me, presumably due to the greater genetic uniformity of a species that mostly self-pollinates (at least for the P. brachystachys; P. aquatica is complicated). I guess DMT and its analogs may be generally more exciting here, though.
I purchased seed from a merchant whose catalog included many entheogenic plants. The merchant gave no information about these seeds beyond their species (where collected from nature, any artificial selection, etc.). I sowed ~20 of each species into respective 3" pots. After the seedlings were big enough to handle, I separated them, selected the most vigorous individuals, and transplanted one seedling each into 1.5" plugs. The potting mix was 50/50 coir/perlite, nutrient profile similar to a cannabis "bloom" or tomato fruiting-stage fertilizer, EC around 2.0 mS/cm, pH around 5.8. A flood table bottom-watered daily, and I hand-watered from the top every week or so to flush out salts. Photoperiod was 16 hours per day under white LEDs, around 7000 lux. These conditions were chosen for convenience, since I already had the system for other plants; I made no attempt to optimize for Phalaris.
Around day 55, I clipped the top ~6" of each P. aquatica, and promptly froze it. Each sample weighed approximately 6 g. A few days later, I chopped each sample coarsely with scissors and placed it in a centrifuge tube. I added stainless steel ball bearings and methanol, and shook the tube with a reciprocating saw for about two minutes. (Bead-beating is widely used in normal research, with purpose-built shakers. I later discovered a Belgian doctor who'd extracted mescaline with similar improvised apparatus[1].) Phalaris leaves are tough, and no amount of beating seemed to fully homogenize them; a stringy cellulosic mat always formed. I wrung that out as best I could, and filtered the liquid under gravity into 100 mL beakers.
For these first six plants, I extracted each sample three times, with three portions of 6 mL methanol each, in a 15 mL tube. I extracted later samples with a single portion of 20 mL methanol, in a 50 mL tube. I don't think that made a big difference. The chlorophyll wasn't fully extracted with the latter, since the water in sample dilutes the methanol to the point that it's not very soluble, but I don't think that affects the alkaloids much.
The beakers were then left to evaporate the methanol and endogenous water. The residue was redissolved in household vinegar, filtered again, made basic with ammonia, and extracted with two portions of 2 mL dichloromethane each, using a long needle on a glass syringe. The dichloromethane was left to evaporate, and the residue was redissolved in 100 uL of methanol. This procedure was loosely adapted from various papers in the seventies.
I spotted these extracts onto silica TLC plates, and eluted with 99:1 methanol to concentrated ammonia. The P. aquatica all showed a single spot under shortwave UV (a mercury lamp sold for algae control in aquariums), which I presumed to be DMT or 5-MeO-DMT. I compared against standards for DMT and 5-MeO-DMT; I think I saw a slight difference in Rf, but not enough to confidently distinguish. Various papers report separation of DMT from 5-MeO-DMT by TLC, but not by much; for example [2] uses HPTLC plates, and the difference is still pretty subtle.
I repeated this procedure for the P. aquatica regrowth anyways, around day 120, with similar results. I also repeated this procedure for the P. brachystachys at the same time. I'd probably left those too long without trimming, since immature seed heads were already present. The P. brachystachys all showed a second well-separated spot, which I assumed (not fully correctly; see below) from its Rf to be gramine. I'd hoped to quantify from the TLC, with an approach similar to [1]'s mescaline work. My results spotting standards were much worse than his though, perhaps because of my generally worse method (cheaper and smaller plates, cruder developing chamber, UV and not stain, etc.). I also found that workflow to be generally cumbersome. I thus became discouraged, since my analysis couldn't distinguish any difference between my plants that would be relevant for breeding.
I thus mostly abandoned the project, leaving the plants in their 1.5" plugs. The P. brachystachys set viable seed and then died around day 200, I guess completing its annual life cycle. A different photoperiod, more frequent trimming, a bigger pot for less dryback, and/or higher nitrogen might have extended its vegetative growth phase and thus yield. The P. aquatica never flowered, but it got hopelessly root-bound in the tiny plug and I eventually trashed it.
Later, I chanced to inherit some HPLC equipment: a single working pump, and a variable wavelength detector (not a DAD, one wavelength at a time). I first tried to develop a method with my inherited C18 column, but was not successful. At low pH, DMT is very weakly retained, even with almost pure water, and the peak tails badly. I probably should have guessed that, since methods in the literature generally use different columns; Clarke's refers to a C8, and [3] (and some others) used a pheny-hexyl. It's possible that a method could be developed on a C18 column at higher pH, but I didn't want to mess with buffers and my column wasn't rated for >8.0.
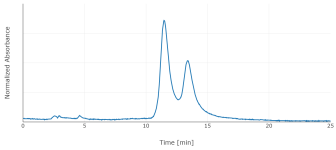
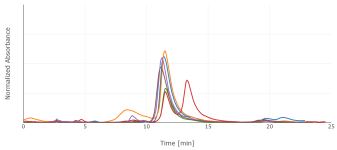
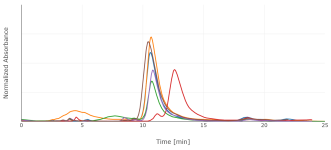
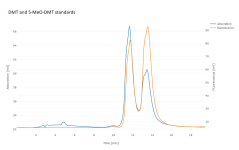
I switched to a phenyl-hexyl column, with 85:15 water:MeOH (w/w) plus 0.1% phosphoric acid. That improved the peak shape somewhat, and separates 5-MeO-DMT from DMT. I'd saved my extracts, though under poor conditions, in microcentrifuge tubes at room temperature. They'd therefore dried out, so I redissolved in 500 uL of methanol, filtered through a syringe filter, and analyzed by HPLC at 275 nm. The P. aquatica shows a fairly simple chromatogram, with DMT as the dominant alkaloid for 5/6 individuals. 1/6 showed both DMT and 5-MeO-DMT. A different extract of multiple seedlings pooled together actually showed more 5-MeO-DMT than DMT.
Those six chromatograms are superimposed here. The sample weights weren't exactly the same, so the chromatograms have been scaled vertically to make that comparable. DMT concentration ranged from 40 to 90 ug/g fresh (not the usual dry) weight. After drying in an oven, a different arbitrary sample decreased in weight by a factor of 8.4.
Don't trust any of these numbers too much. This is with standards of questionable provenance, significantly overlapping peaks, old equipment, etc.; but they did land in the right absolute order of magnitude, and relative comparisons may be more valid. Please also excuse the variation in retention time, which I think is mostly room temperature since I don't have a column oven. It's enough to make correct identification of the peaks somewhat tricky, but I ran standards enough additional times that I'm fairly confident I've done so correctly.
Regrowth showed more DMT than initial growth, 60 to 150 ug/g. This is consistent with the literature, though also consistent with less time for my poorly-stored sample to degrade. The 1/6 plant produced 5-MeO-DMT in both the initial growth and the regrowth.
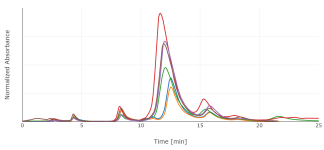
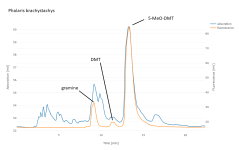
As expected from the TLC, the P. brachystachys shows a more complex chromatogram. We see four additional peaks here at 275 nm, and at least one more at other wavelengths. Gramine is most discussed, but it's not the only unwanted alkaloid and not obviously (at least to me) the most toxic one. 5-MeO-DMT concentration ranged from 100 to 320 ug/g fresh weight.
[2] reviews many alkaloids reported in a different Phalaris species, and develops an HPTLC method to partially separate them. These standards are unfortunately not easily obtained, though. I've re-run the same extract at a few different wavelengths, some of which makes sense (e.g. that only 5-MeO-DMT still absorbs at 310 nm) but most of which is mysterious to me for now. I could get continuous UV spectra by pausing elution and sweeping the wavelength in my detector, though I'm not sure how much that would help.
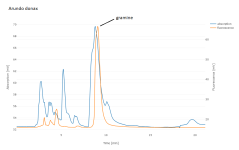
I scraped the "gramine" spot off a TLC plate and found it separated into two peaks by HPLC; so that possibly narrows it down, assuming the sample actually contains gramine and that the gramine is actually eluting. I live near a stand of Arundo donax, which is reported to contain gramine (aka donaxine) as its primary alkaloid, so I may use that for further work.
In any breeding program, repeated testing would be required to determine whether any differences in alkaloid content are consistent. I've seen claims that particular alkaloids may appear and disappear seasonally, and my primary finding (P. aquatica mostly DMT, P. brachystachys mostly 5-MeO-DMT) is exactly backwards from [4].
I'm still considering my next steps. Assuming optimistically the 5-MeO trait in the P.aquatica is recessive and Mendelian, I could probably breed it out in two generations, by selecting DMT-only individuals, then growing out ~30 offspring and confirming those are DMT-only too. (That's just one test, not 30, since I can pool the samples.) I don't know how quickly I could induce flowering in P. aquatica though, so the breeding cycle might be long, and might require more space than I have available indoors. I'd appreciate advice from anyone with experience there.
I can definitely breed the P. brachystachys, with 2-3 generations per year. If my purchased seed is the S0 generation, then I'm now growing S2, after two rounds of selection for maximum 5-MeO-DMT. (I'm indoors with no fan, so I believe self-pollination is much more likely than cross-pollination. I didn't consistently bag the seed heads, though. It would also make sense to select for minimum unwanted alkaloids; but those look constant enough that's effectively the same thing.) I'm simultaneously growing more S0, so that I can compare and determine confidently whether I've actually achieved anything in my breeding. I should be ready to sample again in a month or so.
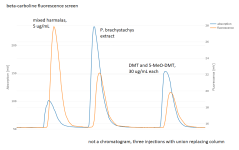
I've also optimized my sample preparation for analysis by HPLC. Sonication seems to work as well as bead beating or better, and it's much less trouble. Acidified water seems to extract about as much analyte as methanol. With detection by UV absorption at any single wavelength, I've been unable to develop any reasonably fast isocratic method that separates DMT and 5-MeO-DMT from the additional unidentified peaks in that crude aqueous extract. SPE is faster than the liquid-liquid extraction used above. I did some initial development there, but then I got a fluorescence detector. That seems to work great; with excitation at a 279 nm and emission at 352 nm, I get response from the DMT and 5-MeO-DMT but nothing else.
So I can process a couple dozen samples in a few hours (plus HPLC time) now, allowing me to grow and select from more individuals. I'll continue my selfing; but at some point I guess even a grass will get too inbred and lose vigor, and I also expect diminishing returns. So I also want to try some crosses. Mechanical emasculation seems difficult for an inflorescence this small. Brief immersion in hot water is reported in [5] to emasculate P. canariensis, which they believe to be a domesticated form of P. brachystachys, so I may try that.
[1]
Validation and exploratory application of a simple, rapid and economical procedure (MESQ) for the quantification of mescaline in fresh cactus tissue and aqueous cactus extracts
[2]
Efficient and Sensitive Method for Quantitative Analysis of Alkaloids in Hardinggrass (Phalaris aquatica L.)
[3]
Synthesis and characterization of high‐purity N,N ‐dimethyltryptamine (DMT) hemifumarate for human clinical trials
[4]
"Ayahuasca-like" effects obtained with Italian plants
[5]
Phalaris canariensis is a domesticated form of P. brachystachys