aizoaceous
Titanium Teammate
This post discusses my experiences with semisynthetic mesembranols. I'm unaware of any prior reports of human use of these drugs in pure form, so note the danger and take due care. I think this was reasonably safe for the reasons discussed below, but it presents greater opportunity for a bad outcome than simple extraction of a known drug.
Mesembrine is an alkaloid extracted from Sceletium tortuousum (syn. Mesembryanthemum tortuosum) and other M. spp. It is usually the most abundant alkaloid, but many structurally similar alkaloids have also been identified as natural products. These include mesembranols, which replace the double-bonded oxygen from mesembrine's carbonyl group with a hydroxyl and a hydrogen. (By analogy, mesembrine is occasionally referred to as mesembranone.) This creates a new stereocenter so two epimers exist, arbitrarily named mesembranol and epimesembranol.
Both epimers occur as natural products, both with abundance low enough that large amounts of plant material would be required to isolate a meaningful quantity. They can also be prepared semi-synthetically from mesembrine purified by chromatography from plant material. I have done so following Patnala's doctoral thesis, which itself follows Jeffs et al. (@Transform - Should I report the details? I know that synthesis talk is sensitive so I've omitted for now. It's a sodium borohydride reduction in methanol. I modified the quench and workup but otherwise just followed the literature. This is legal in most jurisdictions including mine, though would be illegal at least in Lousiana and South Korea.)
The reduction yields a mixture of epimers. Mesembranol base is reported to crystallize from mixed epimers in ethyl acetate. I tried that and got a few tiny crystals, but not enough that I bothered trying to separate them. Patnala reports bigger crystals, but only from a mixture prepared by catalytic hydrogenation that contained relatively less epimesembranol. Jeffs et al. report enough crystals for MS and a melting point from the borohydride product, but don't report the mass. Epimesembranol is a liquid at room temperature.
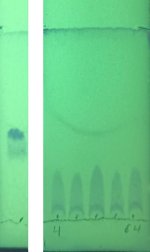
Patnala somewhat unhelpfully reports a TLC only in his usual system of DCM:EtOH, as a long smear. A clean spot is reported by other authors in systems including triethylamine or ammonia. The image below shows the TLC before the reaction, and during the reaction at 4, 8, 16, 32, and 64 minutes. The reaction thus seems to complete in <4 min.
The epimers can be separated by chromatography, but impatience led me to try the mixture first. I prepared eight doses from 5.5 mg to 43 ug by serial dilution by factors of two, each dissolved in 100 uL water as the citrate. I dosed sublingually and held the solution under my tongue for twenty minutes (probably much longer than needed) each time. I waited an hour before increasing the dose each time.
This obviously carries unknown danger and I don't recommend that anyone replicate this. It's less unsafe than a completely novel compound, since if mesembranol were greatly toxic then traditional use of kanna or academic laboratory study of Sceletium extracts (containing natural mesembranols) would have probably revealed that. A permanently harmful overdose of mesembrine seems almost impossible based on animal data. This increases the probability that analogs will be safe, but doesn't guarantee it. Identity of my reaction product isn't absolutely confident without MS or NMR. Reduction of the crude alkaloids without prior purification would yield unknown and potentially dangerous additional products, thus my use of chromatographically purified mesembrine as the starting material. It was already somewhat reckless to consume the product without further purification (by chromatography, crystallization, etc.), and I don't plan to in future.
I felt threshold effects at 170 ug, beginning about 10 minutes after dosing, peaking around 30 min, and mostly over by 50 min. The subjective experience was similar to mesembrine. At higher doses up to 2.8 mg, I got faster onset but similar peak effects and duration. My general impression is that the mesembranols are active at lower dose than mesembrine; but the effects still plateau, and plateau at a level similar to or lower than mesembrine. I experienced moderate nausea at 340 ug, but not at higher or lower doses so probably that was coincidence.
I now have about 200 mg of mixed mesembranol epimers. I may separate those on a flash column, to judge their effects independently. This may help determine whether they hold any interest as drugs, whether semisynthetically like here or as selection targets in Sceletium. I may also try synthesizing esters of mesembranols, which may work as prodrugs. Luche reduction would probably also yield mesembrenols from mesembrenone.
Mesembrine is an alkaloid extracted from Sceletium tortuousum (syn. Mesembryanthemum tortuosum) and other M. spp. It is usually the most abundant alkaloid, but many structurally similar alkaloids have also been identified as natural products. These include mesembranols, which replace the double-bonded oxygen from mesembrine's carbonyl group with a hydroxyl and a hydrogen. (By analogy, mesembrine is occasionally referred to as mesembranone.) This creates a new stereocenter so two epimers exist, arbitrarily named mesembranol and epimesembranol.
Both epimers occur as natural products, both with abundance low enough that large amounts of plant material would be required to isolate a meaningful quantity. They can also be prepared semi-synthetically from mesembrine purified by chromatography from plant material. I have done so following Patnala's doctoral thesis, which itself follows Jeffs et al. (@Transform - Should I report the details? I know that synthesis talk is sensitive so I've omitted for now. It's a sodium borohydride reduction in methanol. I modified the quench and workup but otherwise just followed the literature. This is legal in most jurisdictions including mine, though would be illegal at least in Lousiana and South Korea.)
The reduction yields a mixture of epimers. Mesembranol base is reported to crystallize from mixed epimers in ethyl acetate. I tried that and got a few tiny crystals, but not enough that I bothered trying to separate them. Patnala reports bigger crystals, but only from a mixture prepared by catalytic hydrogenation that contained relatively less epimesembranol. Jeffs et al. report enough crystals for MS and a melting point from the borohydride product, but don't report the mass. Epimesembranol is a liquid at room temperature.
Patnala somewhat unhelpfully reports a TLC only in his usual system of DCM:EtOH, as a long smear. A clean spot is reported by other authors in systems including triethylamine or ammonia. The image below shows the TLC before the reaction, and during the reaction at 4, 8, 16, 32, and 64 minutes. The reaction thus seems to complete in <4 min.

The epimers can be separated by chromatography, but impatience led me to try the mixture first. I prepared eight doses from 5.5 mg to 43 ug by serial dilution by factors of two, each dissolved in 100 uL water as the citrate. I dosed sublingually and held the solution under my tongue for twenty minutes (probably much longer than needed) each time. I waited an hour before increasing the dose each time.
This obviously carries unknown danger and I don't recommend that anyone replicate this. It's less unsafe than a completely novel compound, since if mesembranol were greatly toxic then traditional use of kanna or academic laboratory study of Sceletium extracts (containing natural mesembranols) would have probably revealed that. A permanently harmful overdose of mesembrine seems almost impossible based on animal data. This increases the probability that analogs will be safe, but doesn't guarantee it. Identity of my reaction product isn't absolutely confident without MS or NMR. Reduction of the crude alkaloids without prior purification would yield unknown and potentially dangerous additional products, thus my use of chromatographically purified mesembrine as the starting material. It was already somewhat reckless to consume the product without further purification (by chromatography, crystallization, etc.), and I don't plan to in future.
I felt threshold effects at 170 ug, beginning about 10 minutes after dosing, peaking around 30 min, and mostly over by 50 min. The subjective experience was similar to mesembrine. At higher doses up to 2.8 mg, I got faster onset but similar peak effects and duration. My general impression is that the mesembranols are active at lower dose than mesembrine; but the effects still plateau, and plateau at a level similar to or lower than mesembrine. I experienced moderate nausea at 340 ug, but not at higher or lower doses so probably that was coincidence.
I now have about 200 mg of mixed mesembranol epimers. I may separate those on a flash column, to judge their effects independently. This may help determine whether they hold any interest as drugs, whether semisynthetically like here or as selection targets in Sceletium. I may also try synthesizing esters of mesembranols, which may work as prodrugs. Luche reduction would probably also yield mesembrenols from mesembrenone.
