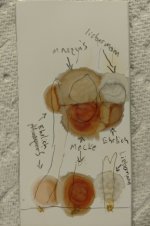
Peganum harmala root extraction:
Due to severe personal time constraints, I have not written this up for a year and a half. hopefully the GCMS results of the samples posted (and received) last year have been returned.
I haven't had the time to work out how to insert images in-line - and hence I am pasting the text below, and hopefully attaching the PDF version with inline images.
Materials and Method
Materials:
Coffee grinder
Dried Peganum harmala roots.
Sodium carbonate
Sodium hydroxide
Sodium chloride (non-iodised rock salt)
Water
Vinegar
Paper towels
Fine tea pot filter
Smooth tea towel
8mm Stainless steel spatula
150mm glass stirring rod
Method:
All steps were accompanied by Tripping Daisy's albums "Bill", "I am an Elastic Firecracker ", and "Jesus Hit Like the Atom Bomb". This is significant, because whenever I listen to this music (which is fairly often) I recall this, my first exploration into the wonders of isolating certain harmala alkaloids from raw herbal material!
Roots were harvested 2-5 years prior in late winter/early spring (when repotting) from container-grown plants grown in potting mix and pumice. The roots were rinsed of most extraneous matter when fresh, then dried in a domestic hot water cupboard. At the time of extraction, there appeared to be some <1mm diameter spherical fungal fruiting bodies on them following storage in a sealed plastic bag.
Root diameter was from ~5mm down to fine root hairs <1mm in diameter. The relative proportion of root diameters was not recorded. The total weight of dried roots was 26g.
The steps outlined in
Harmalas Extraction and Separation Guide - DMT-Nexus Wiki were used as a guideline, as follows:
Roots were powdered in a domestic electric coffee grinder until fine airborne powder was present. Some larger root sections were not ground so finely
The root powder was brought to the boil and simmered lightly in water + vinegar as per the above link <<how much?>> in a stainless steel pot for 15-30 minutes. The liquid was filtered through a fine stainless steel tea filter and the filtered liquid retained. The marc was returned to additional water + vinegar. This process was repeated five times, until there was very little yellow coloration in the liquid.
The acidic aqueous extractions were combined and filtered through coffee filters, paper towels and fine cloth tea towels. This final filtration process took several hours due to the very fine particle size.
Following the final acidified aqueous extraction, the liquid was combined
and reduced to 500ml by gentle simmering, then poured into a 500ml jar.
Sodium carbonate powder was added to the to liquid in small amounts using a small stainless steel lab spatula. Some frothing of solution was observed along with some clouding of the solution.
NaOH was then added to further basify. Litmus paper showed that NaOH definitely increased pH by ~2 points - admittedly difficult to tell with the paper and comparison chart in use.
The liquid was filtered through double-layered coffee filters, and the solids retained. (The filtrate was retained for further basification, labelled "Base #1")
The coffee filters with the filtered solids were placed into 100ml vinegar and hot water added to ~400ml and were vigorous pressed/agitated with a glass stirring rod in order to dissolve all solids.
The liquid was filtered through double-layered coffee filters; although the liquid appeared clear, some dark solids were caught in the filter.
The filter paper and solids were discarded, and the liquid retained for Manske process crystallisation. The total liquid volume was 575ml.
The liquid was heated to simmering, and 69g non-iodised salt was added to the solution, and within 2 minutes was poured into 2 jars to cool to room temperature, and then placed in a refrigerator.
After approximately eight hours of refrigeration, the solution and crystallised solids were filtered through double coffee filters.
The filtered solids were rinsed with the recycled filter liquid in order to concentrate the crystals into the base of the coffee filter. The filters were placed into a hot water cupboard with a toothpick to keep the filter paper open in order to speed the drying of the crystal "mush" in the middle.
Following desiccation the crystals were scraped from the filter paper and weighed. 1.545g of harmala alkaloid salts were recorded.
These solids were dissolved into boiled water. The filter paper was rinsed and the liquid combined.
Sodium bicarbonate was added in order to precipitate (assumedly predominantly) harmine freebase, and left to settle for ~6 hours.
The supernatant liquid was sucked out using a large syringe, then gently decanted. Most of the solids remained settled in the bottom of the flask.
Sodium carbonate was added to the remaining liquid in order to (presumably) precipitate harmaline freebase. There was no apparent clouding of the solution. NaOH was then added to the solution in a further attempt to precipitate harmaline . No localised clouding was observed at all when adding solid NaOH. This reinforces the reports that harmaline is not present in P. harmala roots.
The acidification/basification/Manske process was repeated 4 times. A pale beige freebase powder resulted, with a weight of 1.544g
Based on the above, a yield of ~5.94% of harmala alkaloids of unknown purity and profile appears to have been attained using these materials and method.
The next step is to have this extract analysed, after which conclusions may be drawn and further discussions and recommendations future experiments may be made.
<<Edit 22/3/19: Endlessness has received confirmation from the lab that the extract was analysed as being 51% harmine, no harmaline and the remainder being inactive salts and/or residual water.
This is equivalent to a yield of ~3.02% - call it 3% - harmine from these roots. This confirms previously published reports that the roots contain harmine, but not harmaline, n contrast to the seeds of P. harmala, which contain harmaline and harmine.
Obviously this work needs to replicated in order further elucidate the content of P. harmala roots. It must be pointed out that the intention was to isolate alkaloids that are able to be isolated using Manske NaCl crystalisation.
It is obvious that there were other harmala alkaloids present due to the UV flouresence of the supernatant liquid that was (eventually) discarded after the filtering out of harmine crystals. It would be very interesting to have a raw salt extract from roots analysed to see which other harmala alkaloids are present. This would be especially relevant for those who wish to make a raw tea of the roots for consumption, rather than extracting the harmine: What else is in there? What are the potential benefits be of these other compound? what are the potential risks?