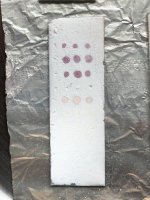
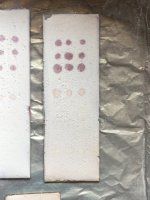
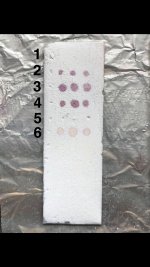
So while preparing solutions for spotting I decided to test 4mg/ml of each powder fraction of different hcl salts of mescaline.
The purist mescaline sample I have been using in all my solubility tests of different salts and even formed a different crystal form has not dissolve what so ever in 96% ethanol. My mescaline carbonate bubbling experiment has had minimal solubility as well which I’d assume means extremely high purity? I’ll be leaving it for 12 hours but even after shaking for a minute zero dissolved.
My mescaline hcl with just normal clean up dissolved partially. And the other “impurities” that are washed all dissolved.
I remember a while back researching MAss spectroscopy and there are different isotopes of the same compound and was wondering if that is another possibility? I have zero idea.
With Cielo being so high yield as well as hplc from altitude perhaps all the salt forms are precipitated since it’ll al be in the curve? I’ll be testing TLC of whatever amount dissolved in the ethanol.
Like I mentioned I have tested these salts separately multiple times and only the more pure mescaline fractions had strong euphoria where the “other alkaloids” which may be mescaline only had energy and higher dose had a “limitless” effect but little to no visuals and little euphoria especially compared to lower doses of the washed white crystals.
1 is my ultra clean mescaline hcl. 2 is the mescaline carbonate which I salted to hcl. The last is some very dark crystals that didn’t precipitate after attempting another round of iso99. These were the impurities that already dissolved earlier.
The purist mescaline sample I have been using in all my solubility tests of different salts and even formed a different crystal form has not dissolve what so ever in 96% ethanol. My mescaline carbonate bubbling experiment has had minimal solubility as well which I’d assume means extremely high purity? I’ll be leaving it for 12 hours but even after shaking for a minute zero dissolved.
My mescaline hcl with just normal clean up dissolved partially. And the other “impurities” that are washed all dissolved.
I remember a while back researching MAss spectroscopy and there are different isotopes of the same compound and was wondering if that is another possibility? I have zero idea.
With Cielo being so high yield as well as hplc from altitude perhaps all the salt forms are precipitated since it’ll al be in the curve? I’ll be testing TLC of whatever amount dissolved in the ethanol.
Like I mentioned I have tested these salts separately multiple times and only the more pure mescaline fractions had strong euphoria where the “other alkaloids” which may be mescaline only had energy and higher dose had a “limitless” effect but little to no visuals and little euphoria especially compared to lower doses of the washed white crystals.
1 is my ultra clean mescaline hcl. 2 is the mescaline carbonate which I salted to hcl. The last is some very dark crystals that didn’t precipitate after attempting another round of iso99. These were the impurities that already dissolved earlier.











 Most things are too 'advance' or just out of my scope since I doubt a college lab would do test for a random citizen considering the compound.
Most things are too 'advance' or just out of my scope since I doubt a college lab would do test for a random citizen considering the compound.