endlessness said:
Thanks Dozuki! Do you write down the Rf's of the spots?
I do occasionally, but I mostly just compare my plates and the relative positions of the compounds. I have images of the plates and can calculate the rF values from the images easy enough. I don't use analytical grade solvents so my rF values are going to be a little different than the literature. For example I use 30% AcOH as opposed to 99% as that is what I have. Comparative positions and color reactions to UV, and spray reagents help to further indicate possible substances. I just received some silicotungstic acid, so I will soon add that to the repertoire. And I wan't to start with UV-Viz here soon.
Dozuki, whats your take on the phenetylamines, arent you surprised by that too?
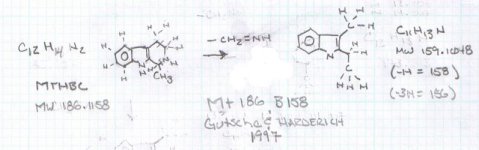
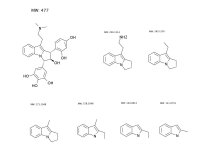
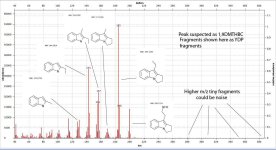
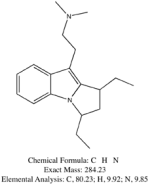
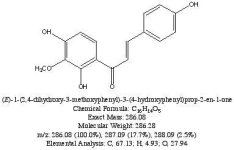
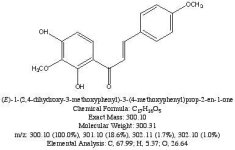
Not really. Hordenine, which is only one hydroxy group away from N,N-dimethyl-phenethylamine is found in Mimosa ophthalmocentra. Again, I would suspect these are degradation products and link back into tannins. My thinking here is that tannins complex with DMT in the plant to produce yuremamine (for the plants and the plant parts that produce it). Under extraction conditions yuremamine breaks down into degradation products that resemble the tannins and tryptamines. I suspect what it breaks down to is highly variable upon the extraction conditions. The phenethylamines could be one of these YDPs. The N-methyl-phenethylamine MS looks like a good match, however, the 2nd one seems 'off'. This would seem to indicate the possibility of being a weird degradation product. There are other spectra in all of these analyses that show the same base peak (185 for instance) but then have different spectra such as T-2 and some of the suspected beta-carbolines. They have some similarities (m/z 185, 144, 113-114) but then have totally different M+. Seems like it might be one compound breaking down in various different ways.
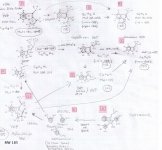
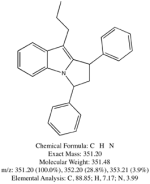
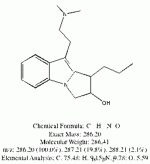
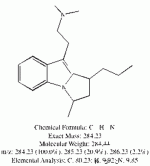
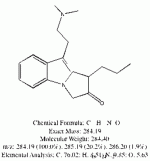
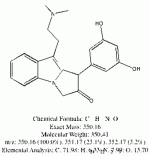
I spent the evening drawing variations on what the break down products of yuremamine might be and figuring out the molecular weight. I keep going back to the quote in the original 2005 yuremamine paper: "Unidentifiable degradation products". So far I haven't been able to match anything up precisely. Inside the molecule of yuremamine, you can see N-methyl-penethylamine, and N,N-dimethyl-phenethylamine, DMT, NMT, and phenol based compounds related to the tannins. It's all in there, it just seems to be a big cosmic puzzle as to how it comes out.
Realize however, that this is still my working *hypothesis*. It was borne of an 'AH HA!' moment that I had when reading that original 2005 paper after being highly puzzled over the disappearing spot #1.
I thought that the NMT spot was one of the degradation products as well. At that time the consensus thought was that MHRB was a very pure source for DMT. There were really only a couple papers on it (known), one of which claimed it was the sole alkaloid (via paper chromatography). The work being done here is showing compounds like NMT and the beta-carbolines in MHRB where that has not really been show before. I think some of these other compounds are being produced by the plant, but we have to sift through what is a natural product of MHRB and what are degradation products, and analysis artifacts (DMT oxide for example).
I think there is publishable data here. How to go about it is another thing. What publication? Above ground or underground publication? For any above ground scientific journals you might have to compare your results with reference samples. Also, the identification of the starting material might have to be more precise. Getting root bark from a middle man vendor, its impossible to know the true identity of the species being looked at is. With the work for Acacias, this might be a little easier as you could obtain a specimen. MHRB specimen might be another story, I don't know.
I think that its awesome that results are getting published here on the Nexus! That in itself is publication. The only reason that I found The Nexus was because Entropymancer used some of my plates in the paper he got published in The Entheogen review. I discovered that via Google one day because I was curious what ever became of the whole yuremamine/jungle spice mystery and I read his drafts of that paper here.
Either way, this is ground breaking stuff that I feel privileged to be involved with.