downwardsfromzero said:
I've probably mentioned in this thread already that a simple method has been suggested which exploits the good solubility of psilocybin in hot water and the phosphate group's ability to bind to calcium: precipitate the psilocybin from hot water using a calcium salt. Boiling will also degrade any unfavourable enzymes.
I tried this once although life happened before I got a chance to thoroughly investigate the result.
Powdered P. allenii ('cyanofriscosa') is so insanely potent that it's not worth the trouble of extracting

Any updates on the hunt for the Ca salt of psilocybin? Seems like it is worth pursuing further.
Here is how I see the situation (all this is speculation):
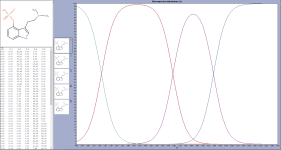
1) We would want psilocybin to have a -2 charge when adding Ca. This is because if it is at -1 charge, then only monocalcium is obtainable and it may remain soluble (otherwise precipitation of psilocybin should be simple). To get to the -2 charge we need a pH of 12 or above (see first image courtesy of Ulim).
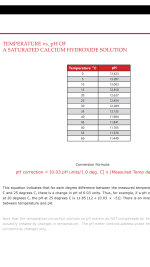
2) At very high pH there is the problem that insoluble Ca(OH)2 will form and precipitate instead/before our desired salt. Indeed, Ca(OH)2 becomes insoluble around pH 12 at room temperature (see second image). Process window is tiny and theoretical, not an ideal situation.
3) So the key here may be to find a window where Ca(OH)2 precipitation is not an issue, while at the same time having psilocybin in a -2 charge state. At room temperature this seems difficult, but fortunately Ca(OH)2 solubility increases with lower temps. Near freezing it remains soluble even at pH 13 (second image).
It looks like there may be a process window to precipitate the salt we want if we control the pH and temperature. Procedure would be something like this:
1) Perform basic hot water extract on mushrooms (pH ~ 8 ). Suggesting to keep extract slightly basic to avoid conversion into psilocin through enzymatic action, which could be an issue for cubensis based on the previous posts in this thread.
2) Bring temperature down to near freezing (O C). Allow to settle and filter.
3) SLOWLY and while keeping extract near freezing add small amounts of Ca(OH)2 keep track of pH and what precipitates. Once pH gets to ~13.5 we are done as Ca(OH)2 will no longer dissolve and become an unwanted precipitate itself. We are looking/hoping for a window where the phosphate psilocybin dicalcium salt precipitates first before Ca(OH)2 which is why we lowered the temperature.
4) Collect precipitate(s) and water wash until water comes out neutral. Note that different precipitates may form at different pH. Precipitates (if they form) should contain all kinds of biological phosphate salts, hopefully psilocybin is part of them, and it may be last one to crash out since other phosphate salts seem to crash out readily (see for example the beet sugar refinement
process).
In a twist, Mg can also be added. It seems that Ca/Mg/Phosphate salts are very insoluble (see first attachment where apatites are discussed). CaCl2 may also be helpful.
Any thoughts? Obvious reason(s) this will not work? Has this already been tried without success? Is something like this already known/used? If no feedback I plan to try this later.