Ah, OK - I've drawn out the possible mechanisms, regardlessThe salt was formed from a tea with zero alcohol. The issue that may have happened is the pka of carbonic acid is too low to be 'freed' by the hcl. I just checked and the other mescaline hcl that is from my recycled salting experiments was also carbonate salt so two instances that show hcl doesn't replace the carbonate. I considered the pka to be 6.35 but anhydrous is lower.
The alcohol is the experiment I'm gonna test since seems that the salt is also insoluble in ethanol 96%. Even now after 2 hours it didn't dissolve. Unless it is a question of purity? The salt from from the tea using co2 is the expected amount of mescaline I get after the iso99 washes to remove color. IME the 'pure mescaline' has represented 25-35% of the total alkaloids (ONLY once did it represent nearly entire alkaloid content for me and might have just been error on my end)

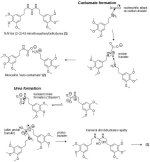
Maybe the "carbonate" has at least been the carbamate all along, if not the urea. Consider how unsubstituted or monosubstituted tryptamines readily form carbamates with CO2, as in the separation methods for NMT.








