downwardsfromzero said:
It looks much like an isostere of 5-MeO-2-Ph-DMT, and more easily approachable to boot.
It also posses very similar pharmocology to 5-methoxy-2-phenyl-N,N-dimethyltryptamine and 2-ethyl-5-methoxy-N,N-Dimethyltryptamine (EMDT)
This is actually turning out to be a rich field of study.
I'm just beginning to research these novel tryptamine compounds which interact with the serotonin 6 receptor, so I have a good deal of work to do, but as I learn and understand more I will continue to add to This thread
Below, in the sectioned off areas, some miscellaneous related research can be found:
-------
Optically active pyrrolidinylmethylindole analogs related in structure to the benzenesulfonyltryptamine 5-HT(6) receptor antagonist MS-245 were evaluated and their R-isomers were found to bind with affinity higher than their S-enantiomers.

www.ncbi.nlm.nih.gov
Interaction of chiral MS-245 analogs at h5-HT6 receptors
------
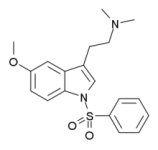
5-HT(6) serotonin receptors are distributed within some dopamine terminal regions in the brain leading to suggestions that they might influence dopaminergic function. In the present study, the 5-HT(6) antagonist 5-methoxy-N,N-dimethyl-N(1)-benzenesulfonyltryptamine (MS-245) was without effect...

www.ncbi.nlm.nih.gov
Abstract
5-HT(6) serotonin receptors are distributed within some dopamine terminal regions in the brain leading to suggestions that they might influence dopaminergic function. In the present study, the 5-HT(6) antagonist 5-methoxy-N,N-dimethyl-N(1)-benzenesulfonyltryptamine (MS-245) was without effect when administered (3.0-7.5 mg/kg) to rats trained to discriminate (+)amphetamine (1.0 mg/kg) from saline vehicle in a two-lever drug discrimination task. Administered in combination, 0.3 mg/kg (i.e., the ED(50) dose) of (+)amphetamine plus 5.0 mg/kg of MS-245 elicited 95% amphetamine-appropriate responding. Similar studies were conducted using rats trained to discriminate cocaine (8.0 mg/kg) from saline vehicle, but a combination of 2.0 mg/kg (i.e., the ED(50) dose) of cocaine together with relatively low doses of MS-245 resulted in the percent response (approximately 50%) expected from administration of this dose of cocaine or in disruption of the animals' behavior. The present results confirm findings from other laboratories that 5-HT(6) antagonists can modulate amphetamine-induced behavioral actions, and further extend these findings to an example of a different structural class of 5-HT(6) antagonists and to a different behavioral paradigm. Taken together, the data suggest that 5-HT(6) serotonin agents (or at least MS-245) could have potential clinical application in therapies that involve modulation of dopamine neurotransmission.
------
Binding of serotonin and N1-benzenesulfonyltryptamine-related analogs at human 5-HT6 serotonin receptors: receptor modeling studies.
Abstract
A population of 100 graphics models of the human 5-HT6 serotonin receptor was constructed based on the structure of bovine rhodopsin. The endogenous tryptamine-based agonist serotonin (5-HT; 1) and the benzenesulfonyl-containing tryptamine-derived 5-HT6 receptor antagonist MS-245 (4a) were automatically docked with each of the 100 receptor models using a genetic algorithm approach. Similar studies were conducted with the more selective 5-HT6 receptor agonist EMDT (5) and optical isomers of EMDT-related analog 8, as well as with optical isomers of MS-245 (4a)-related and benzenesulfonyl-containing pyrrolidine 6 and aminotetralin 7. Although associated with the same general aromatic/hydrophobic binding cluster, 5-HT (1) and MS-245 (4a) were found to preferentially bind with distinct receptor conformations, and did so with different binding orientations (i.e., poses). A 5-HT pose/model was found to be common to EMDT (5) and its analogs, whereas that identified for MS-245 (4a) was found common to benzenesulfonyl-containing compounds. Specific amino acid residues were identified that can participate in binding, and evaluation of a sulfenamide analog of MS-245 indicates for the first time that the presence of the sulfonyl oxygen atoms enhances receptor affinity. The results indicate that the presence or absence of an N1-benzenesulfonyl group is a major determinant of the manner in which tryptamine-related agents bind at 5-HT6 serotonin receptors.
------
Almost two years ago, we launched PubMed Journals, an NCBI Labs project. PubMed Journals helped people follow the latest biomedical literature by making it easier to find and follow journals, browse new articles, and included a Journal News Feed to track new arrivals news links, trending...

www.ncbi.nlm.nih.gov
Abstract
N-Benzenesulfonyl-5-methoxy-N,N-dimethyltryptamine (BS/5-OMe DMT; 5) was shown to bind at human 5-HT6 serotonin receptors with high affinity (Ki = 2.3 nM) relative to serotonin (Ki = 78 nM). Structural variation failed to result in significantly enhanced affinity. BS/5-OMe DMT acts as an antagonist of 5-HT-stimulated adenylate cyclase (pA2 = 8.88 nM) and may represent the first member of a novel class of 5-HT6 antagonists.
-------
-eg